The Global Authority for Regenerative Medicine & Stem Cell Innovation.
One destination. Infinite breakthroughs. Integrating the world's first AI-grounded research hub with a 365-day virtual expo and global summit.
AI Research Navigator
Instant access to the world's stem cell knowledge base, grounded in peer-reviewed literature.
Powered by SCN Research AI · Sources: PubMed, ClinicalTrials.gov, FDA
Innovation Store Spaces
Explore cutting-edge exhibitors, from biotech labs to device providers, available year-round.

NeuroGen Therapeutics
Pioneering neural stem cell therapies for neurodegenerative disorders with proprietary differentiation protocols.

MedDevice Innovations
Next-generation bioreactors and cell expansion systems for scalable stem cell manufacturing.

Pacific Regenerative Clinic
FDA-registered facility specializing in autologous stem cell treatments for orthopedic conditions.

CellCulture Dynamics
Advanced cell culture media and growth factor optimization for mesenchymal stem cells.

PharmaRegen Labs
Drug development pipeline focused on small-molecule enhancers of endogenous stem cell repair.

CRISPR Stem Solutions
Gene-edited iPSC platforms for personalized regenerative medicine and disease modeling.

OrthoStem Devices
Biocompatible scaffolds and 3D-printed implants seeded with patient-derived stem cells.
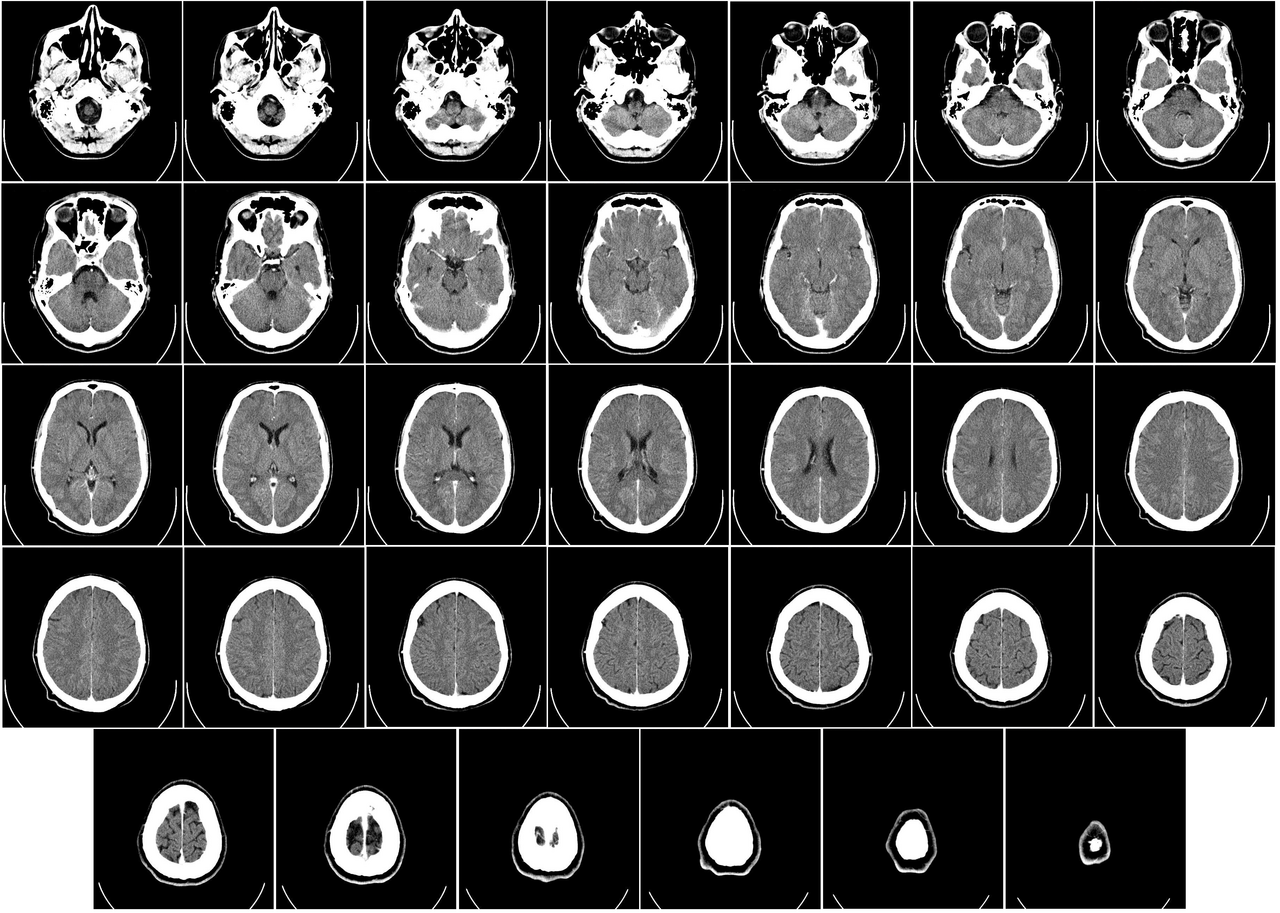
NeuroScan Imaging
Advanced neuroimaging and AI-assisted diagnostics for tracking stem cell therapy outcomes.

Apex Clinical Research
Multi-site clinical trial management for Phase I–III stem cell therapy studies.

StemVault Cryogenics
Cryopreservation and biobanking services ensuring long-term viability of stem cell lines.

RegenFlow Systems
Microfluidic platforms for real-time stem cell sorting, analysis, and quality control.
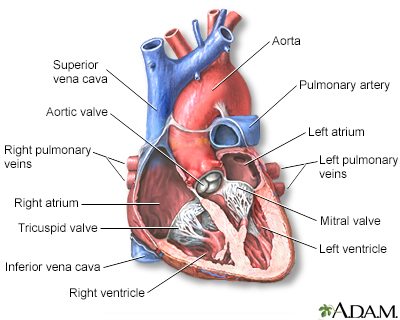
Pinnacle Stem Cell Clinic
Patient-centered regenerative care with focus on cardiac and pulmonary stem cell therapies.
Live Community Feed
Join the conversation with researchers, clinicians, and innovators shaping the future of regenerative medicine.

Ethics of CRISPR in Stem Cells
The recent FDA guidance on gene-edited stem cells raises important questions about germline modifications...

Funding for Phase II Trials
Securing bridge financing between Phase I success and Phase II enrollment remains the valley of death for many...

iPSC Manufacturing at Scale
Our team achieved a 10x improvement in iPSC yield using a novel suspension bioreactor configuration...

Exosome Therapy: Hype or Hope?
The distinction between stem cell-derived exosomes and whole-cell therapies needs clearer regulatory frameworks...

Cardiac Repair Breakthroughs
New data from the REGEN-HEART trial shows a 23% improvement in ejection fraction at 12-month follow-up...

Regulatory Pathways in the EU vs US
The EMA's ATMP classification creates both opportunities and hurdles that differ substantially from FDA's RMAT...
Medical Advisory Board
Guided by world-class experts in regenerative medicine, stem cell biology, and clinical research.

Dr. Richard Harmon
Chief Scientific Advisor
MD, PhD — Harvard Medical School

Dr. Sarah Chen
Director of Clinical Research
MD — Stanford University

Prof. James Whitfield
Regulatory Affairs Lead
PhD — Johns Hopkins University

Dr. Amara Okafor
Head of Translational Medicine
MD, PhD — MIT

Dr. Michael Torres
Orthopedic Regeneration Specialist
MD — Mayo Clinic